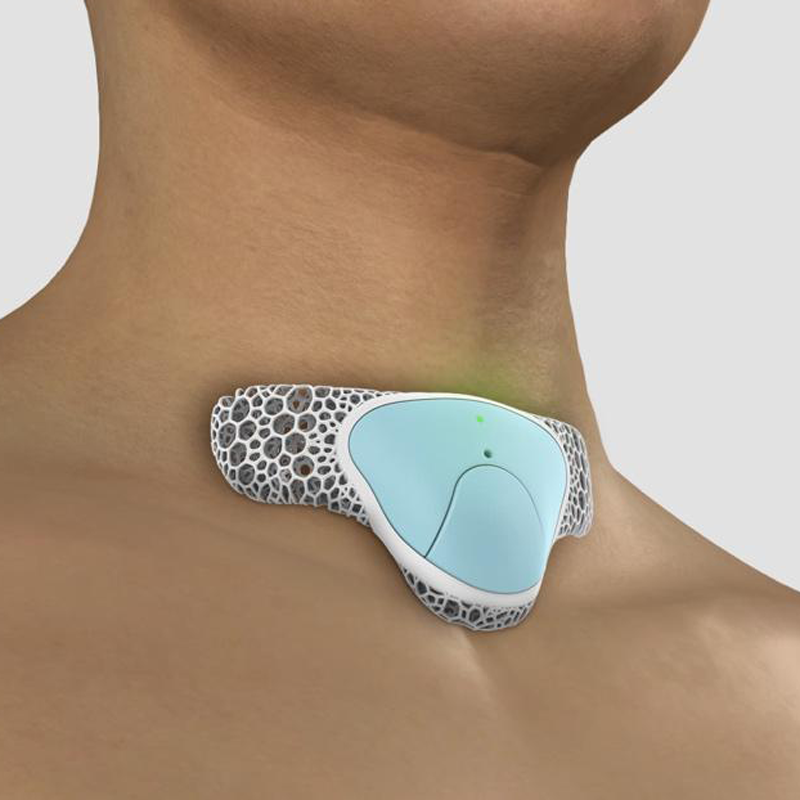
The RTM Respiratory Monitoring System (RMS) is a revolutionary, human-centric, AI-driven respiratory monitoring device that fits comfortably on any user.
It is a small and powerful wearable device ergonomically designed to address the critical need for complete and continuous respiratory monitoring.

RTMpod | Electronic Device
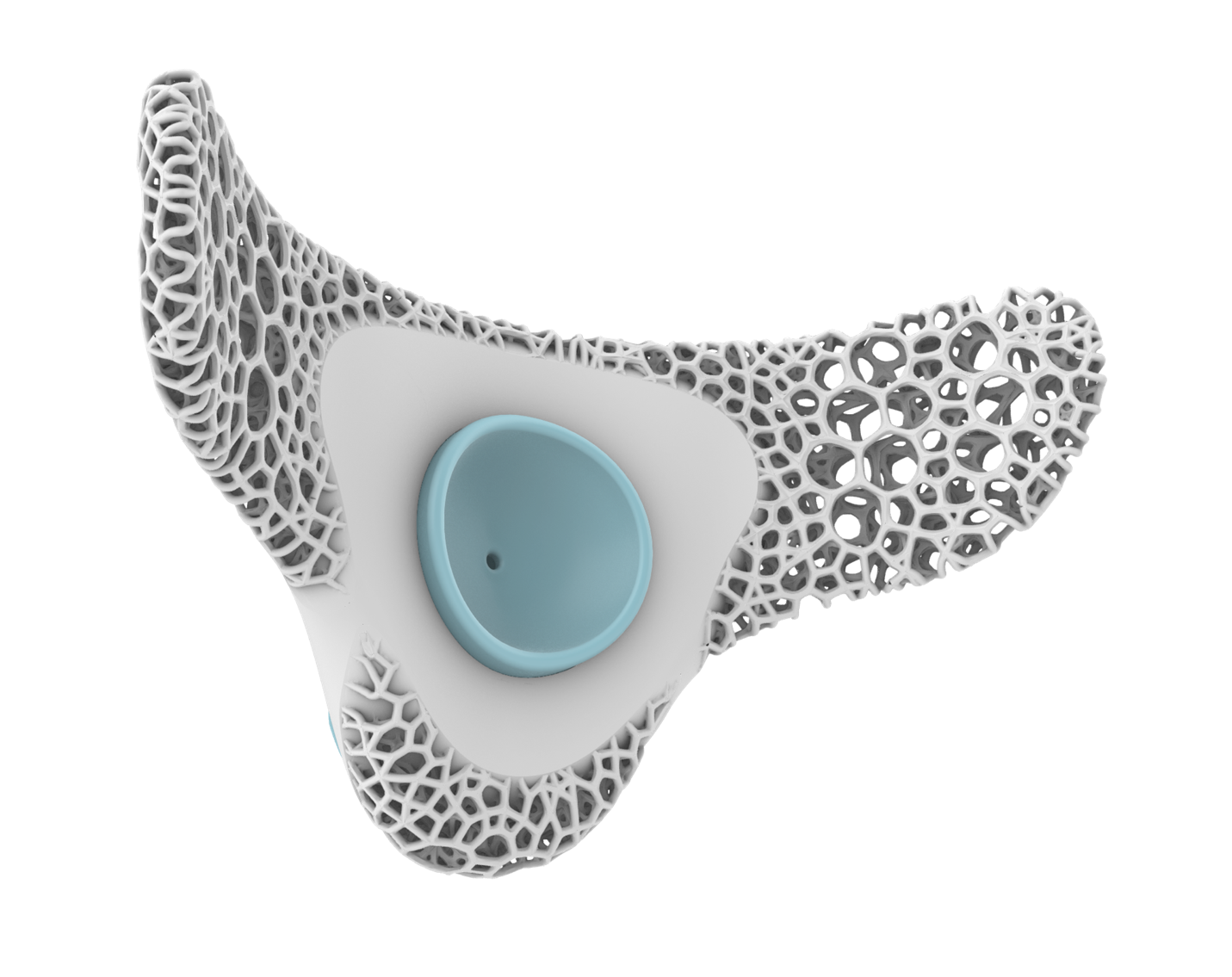
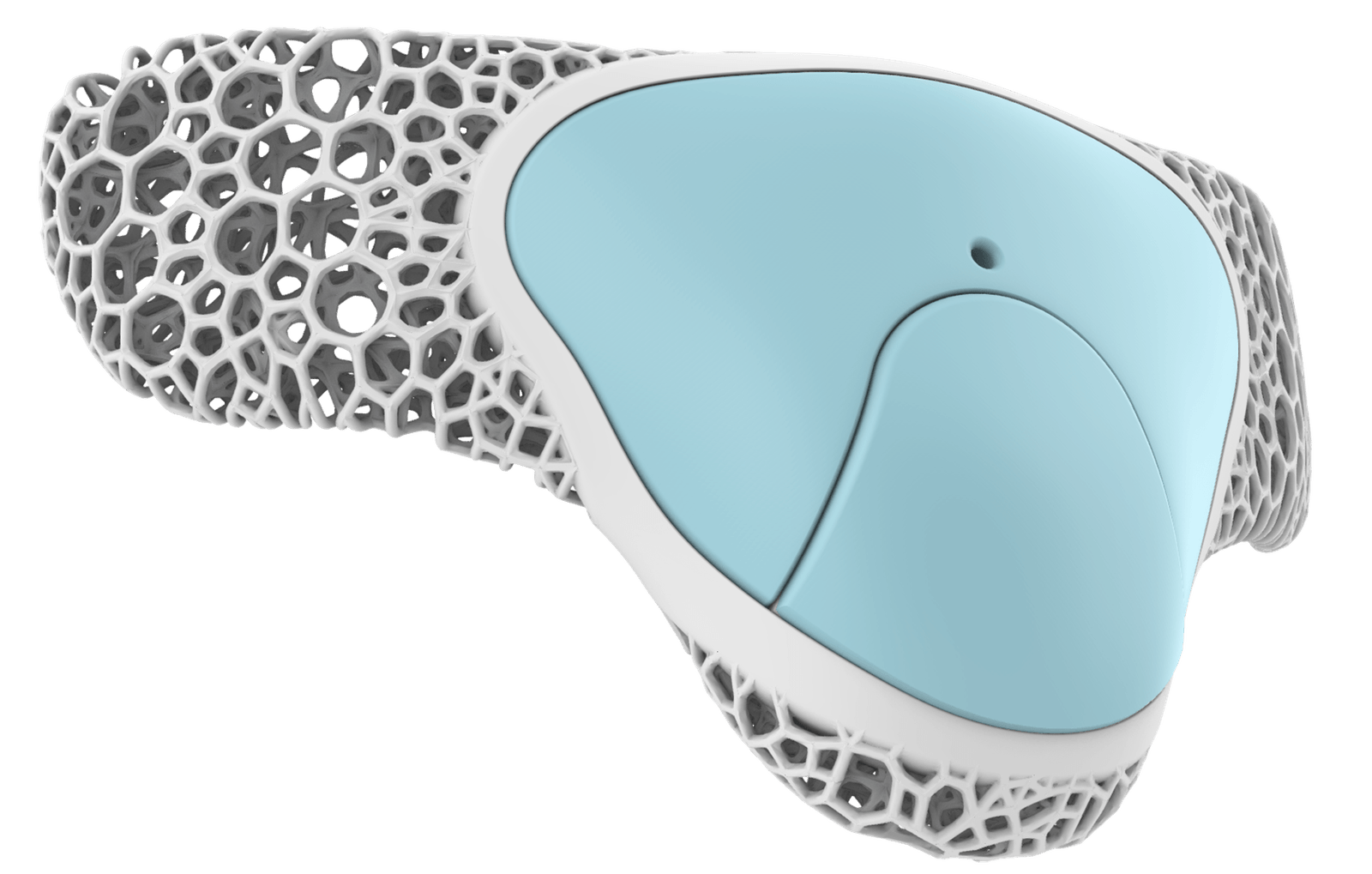
RTMpod is the main electronics device with the audio sensory, onboard processing, and wireless communication capability.
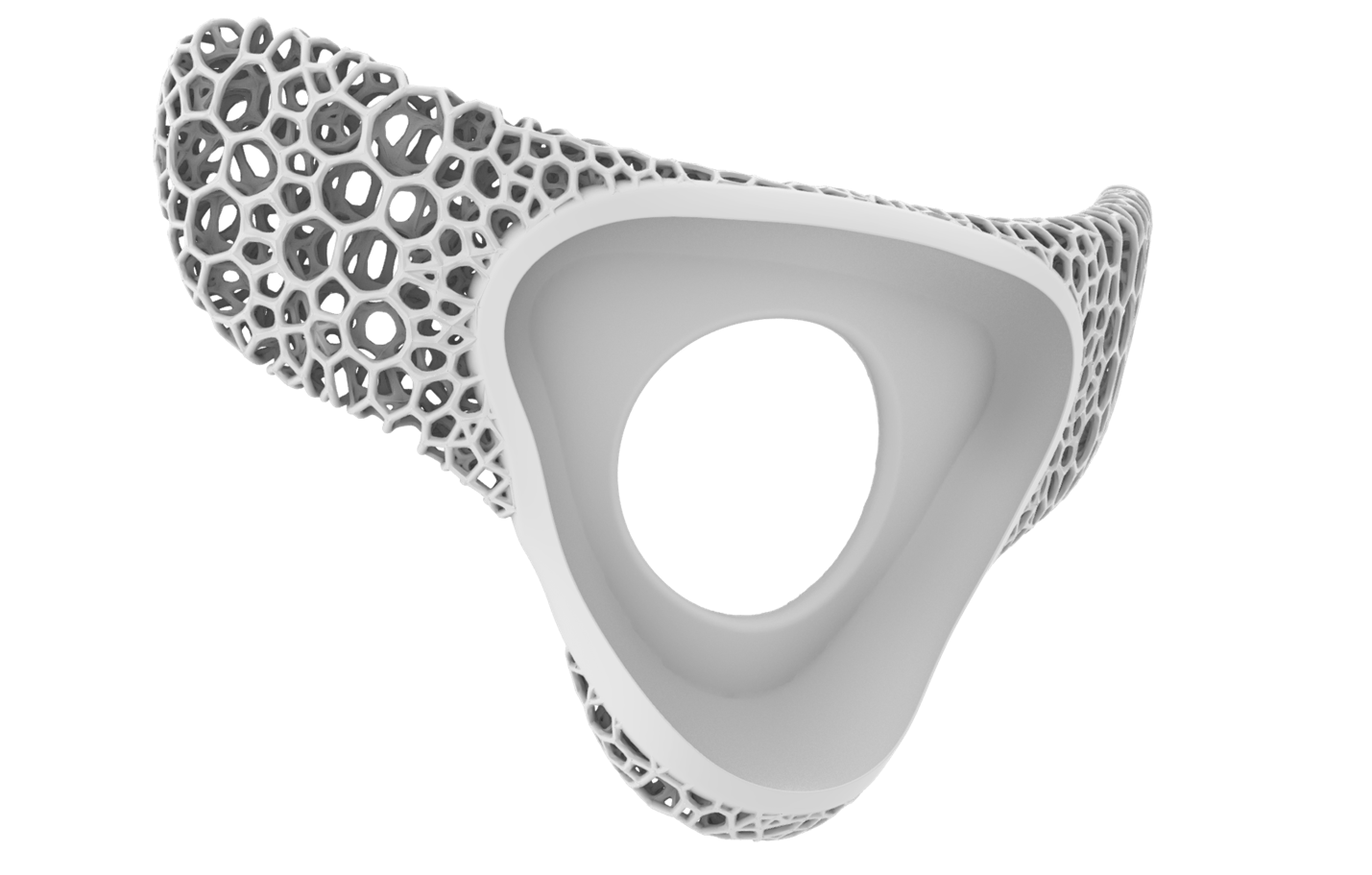
RTMwear | Wearable Device Carrier
The RTMwear is a disposable carrier for the RTMpod and is worn by the patient for the duration of their monitoring
The RTMwear is sold in sealed replenishment packs.
Small, flexible, and lightweight, designed to be comfortable and breathable for any user.
Stays adhered to the patient for up to 72 hours, allowing the RTMpod to be easily removed and replaced.

RTMpack | Rechargeable Battery
Each RTMpack provides up to ten hours of charge and can be easily swapped out with a recharged pack for minimum monitoring interruption.
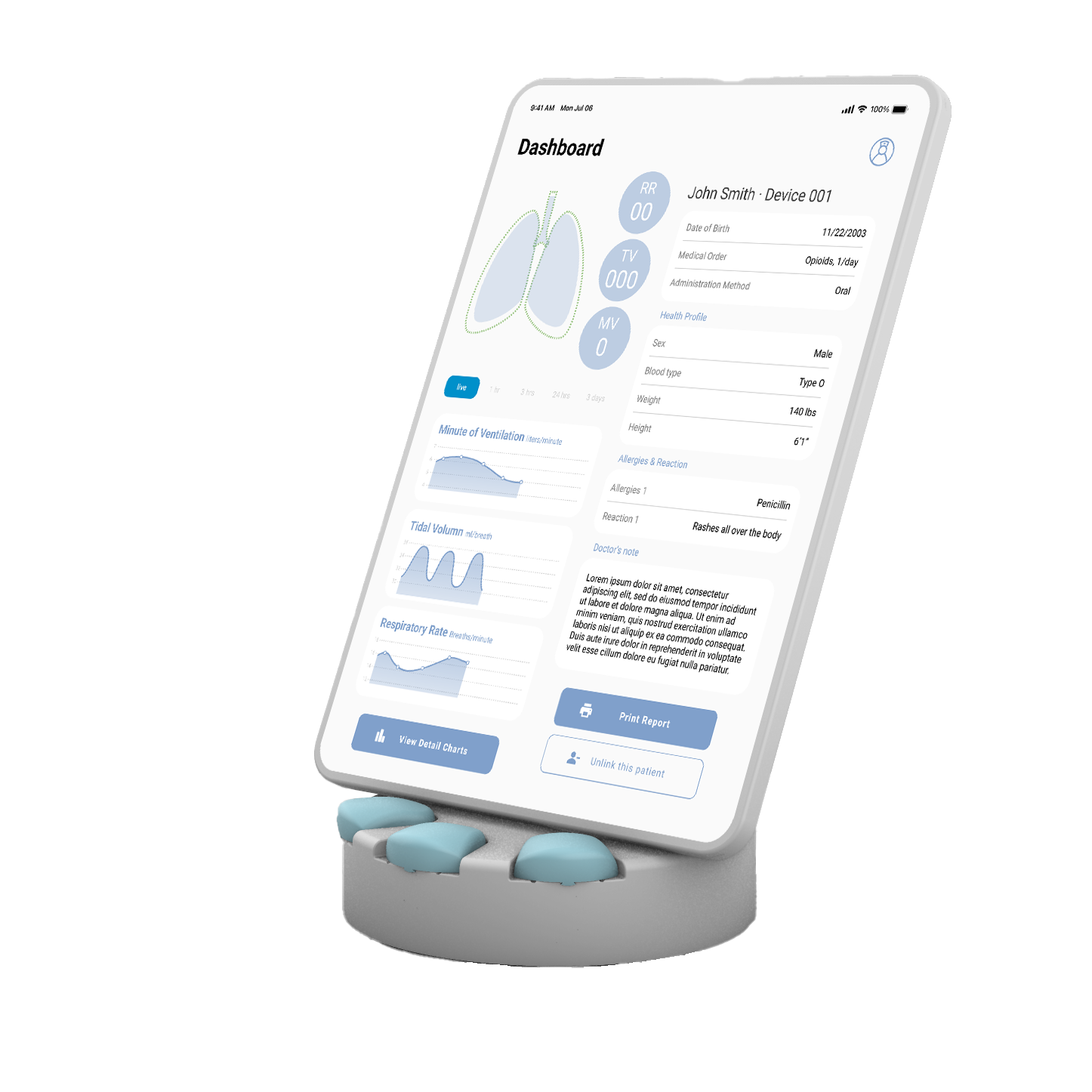
RTMhub | Monitor & Charging Hub
The RTMhub is the main hub for the product system with three key functions:
The RTMhub is the pairing hub for the monitoring device and maintains consistent local communication and data transmission from the device to the backend cloud platform.
The RTMhub provides data display and other information on a tablet for patients, nurses, and doctors.
The RTMhub is also a charging dock for the replacement batteries.
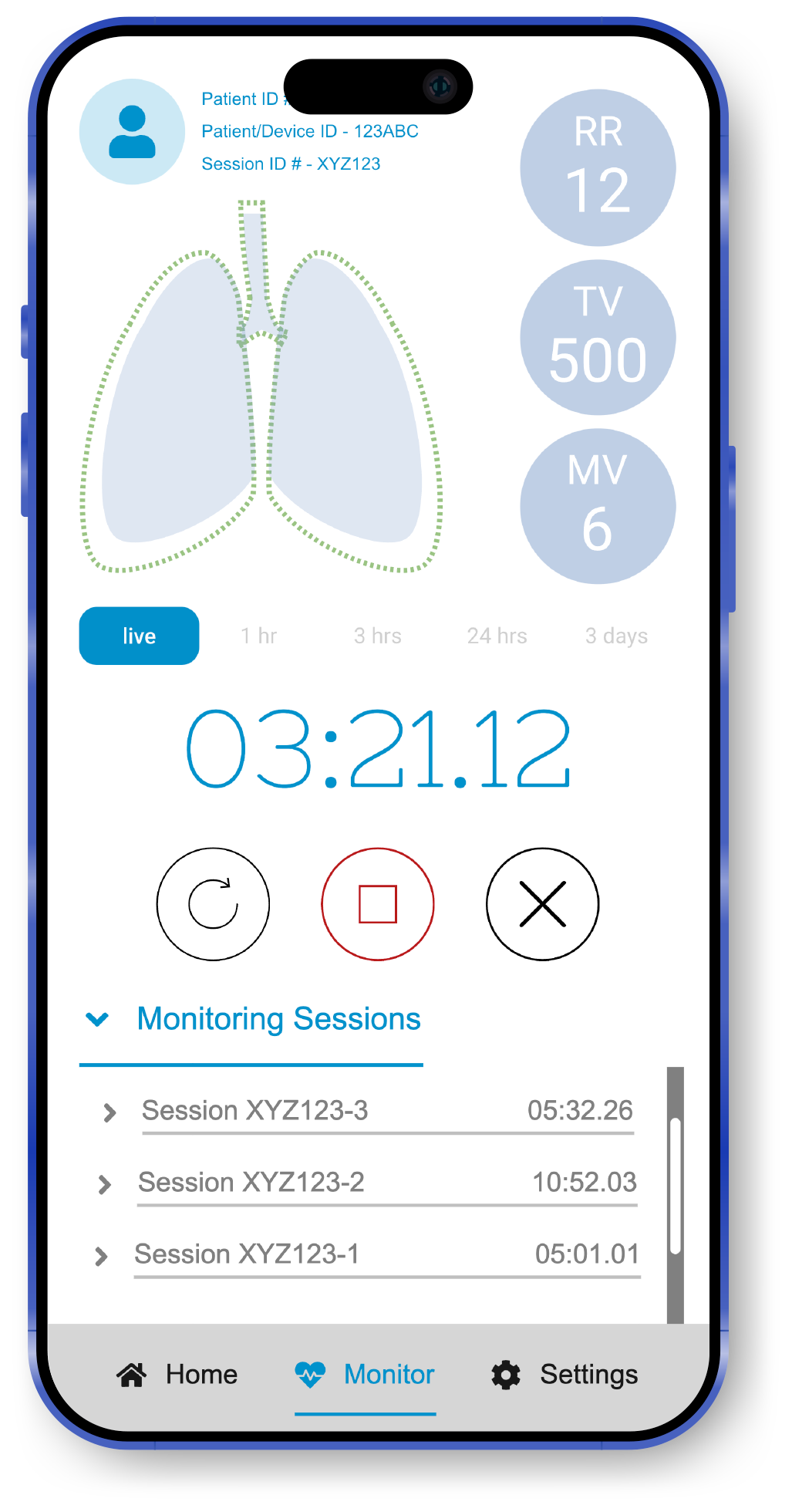
RTMpulse | Multi-User Patient Monitoring Services
Digital services provide any user with access credentials to monitor a patient’s session remotely and receive status notifications.
Depending on the user’s profile (Admin, Doctor, Clinician, Patient, or Caregiver), the associated software presents a different set of data & information contextual to the user’s needs and access clearance.
Ergonomically designed wearable with all user’s needs and situations in mind
Small & lightweight form factor
Flexible material
Comfortable & breathable
Easy application & removal

Ultra-sensitive stethoscope easily positioned and comfortably placed and securely held on a user’s trachea
Real-time tidal volume data
Real-time respiratory rate
Calculated minute ventilation
Connected & wireless technology that is barely felt by the user while providing:
Accurate & continuous respiratory data
Pattern & trend analysis
Predictive algorithms
Real-time alerts

RTM’s Accomplishments and Patented Product Technology & Design
FDA Breakthrough Device Designation
2 human clinical trials completed:
- NIH Funded trial with hospitalized patients managed with opioids
- RTM-funded pulmonary lab study with volunteers
Data used to optimize sensors, usability, software, and algorithms
Human studies demonstrated the ability to measure TV, RR, and HR. The algorithm analyzes trends and patterns of respiratory data
10 US Patents issued, and several pending

RTM has been chosen as a case study for MIT’s Entrepreneurship Bootcamp
RTM received the First Prize award for innovation at the 2023 Anesthesiology Conference
RTM has been awarded 2 NIH Grants

